
电话:+86-755-86967710
邮箱:webmaster@szbl.ac.cn

Institute of Infectious Diseases
chenmx(at)szbl.ac.cn
Pathogenic Infection and Immune Responses,Immune Responses and Metabolism,Immunology,Microbiology
Junior Principal Investigator
Postdoctoral Fellow
PhD
Bachelor
Our research aims to identify biological targets for the prevention and treatment of infectious diseases. The laboratory focuses on understanding the pathogenic mechanisms of microbial pathogens within the host and elucidating how host immunity and cellular metabolism defend against infection at the molecular level.
Our major research directions include:
1. Pathogenic mechanisms of microbial pathogens
2. Molecular mechanisms of host defense against microbial infection, with an emphasis on immune and metabolic regulation
3. Discovery and characterization of novel antimicrobial molecules
We foster a collaborative, supportive, and intellectually stimulating research environment where members work together and grow together. We warmly welcome students and researchers who are interested in infection biology, immunology, and metabolism to join our group.
Dr. Meixin Chen joined Shenzhen Bay Laboratory as a full-time Distinguished Research Fellow at the Institute of Infectious Diseases in 2021 and serves as a PhD supervisor. Dr. Meixin Chen has long been dedicated to studying the interactions between host immunity and pathogen infection. Dr. Meixin Chen’s research first revealed the important role and regulatory mechanisms of ubiquitination of the DNA sensor cGAS in host defense against pathogen infection. In addition, Dr. Meixin Chen was among the first to use metabolite-sensing reporters to uncover an itaconate-mediated antibacterial pathway in host cells. These studies have provided important insights into the mechanisms of pathogen virulence and host defense, contributing to a deeper understanding of host-pathogen interactions. Dr. Meixin Chen has published as corresponding author or first author in leading journals, including Science, Molecular Cell, Nature Microbiology, Advanced Science, and Nature Communications. Dr. Meixin Chen has also received funding from national, provincial, and municipal research programs.
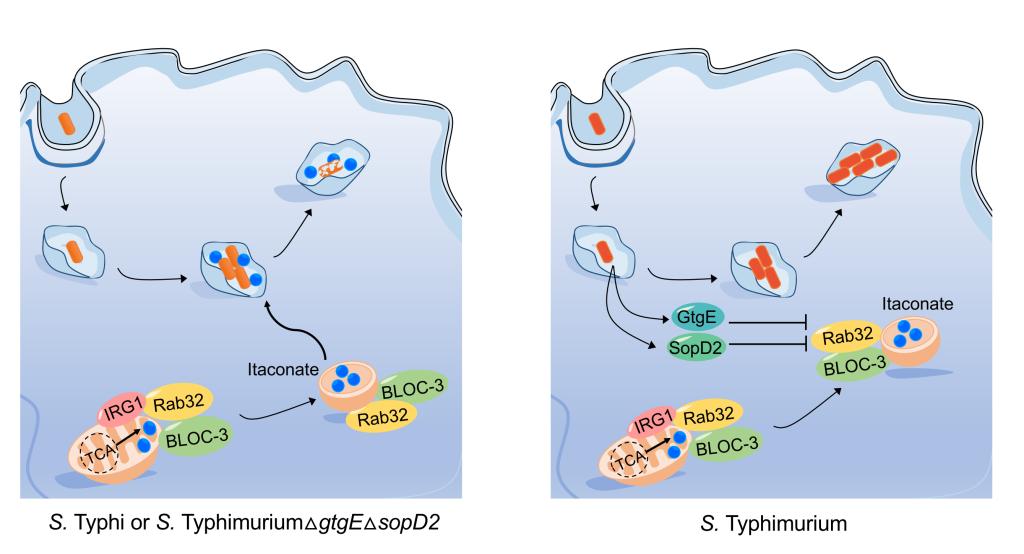
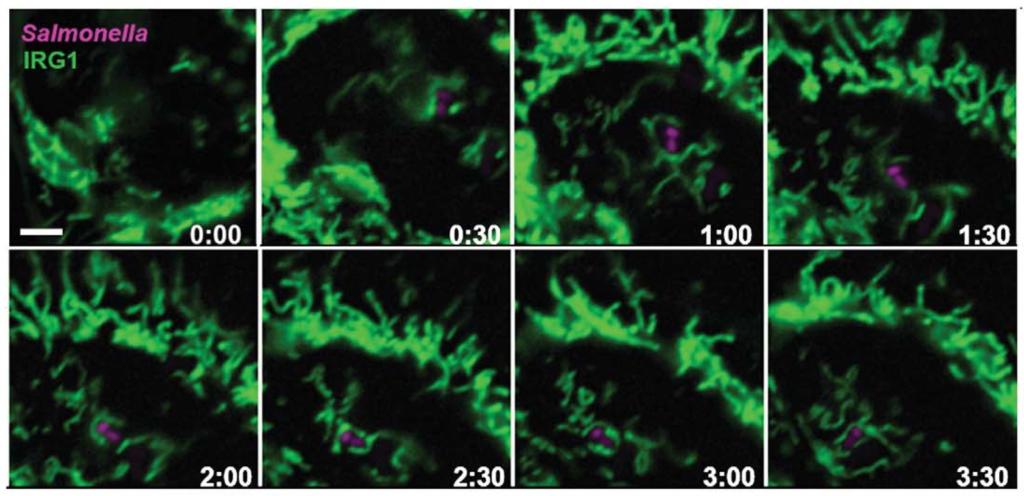
The antibacterial pathway of mitochondrial itaconate
Meixin Chen, et al. Science 369.6502 (2020): 450-455.
1. Meng, Q.#, Li, C.#, Cai, Y., Chen, Y., Chen, X., Wang, X., Zhang, B., Zhang, Y., Liu F., Chen, M*. (2025). Itaconate transport across the plasma membrane and Salmonella-containing vacuole via MCT1/4 modulates macrophage antibacterial activity. Nature Communications, 16 (1), 10551.
2. Chen, M.#, Sun, H. #, Boot, M., Shao, L., Chang, S.J., Wang, W., Lam, T.T., Lara-Tejero, M., Rego, E.H. and Galán, J.E. *, 2020. Itaconate is an effector of a Rab GTPase cell-autonomous host defense pathway against Salmonella. Science, 369(6502), pp.450-455.
3. Lian, H. #, Park, D. #, Chen, M.,Schueder, F., Lara-Tejero, M., Liu, J., & Galán, J. E*, 2023. Parkinson’s disease kinase LRRK2 coordinates a cell-intrinsic itaconate-dependent defence pathway against intracellular Salmonella. Nature Microbiology, 1-16.
4. Chen, M.#, Zhao, Z. #, Meng, Q. #, Liang, P. #, Su, Z., Wu, Y., Huang, J. and Cui, J. *, 2019. TRIM14 Promotes Noncanonical NF‐kB Activation by Modulating p100/p52 Stability via Selective Autophagy. Advanced Science, p.1901261.
5. Chen, M.#, Meng, Q. #, Qin, Y. #, Liang, P., Tan, P., He, L., Zhou, Y., Chen, Y., Huang, J. *, Wang, R.F. * and Cui, J. *, 2016. TRIM14 inhibits cGAS degradation mediated by selective autophagy receptor p62 to promote innate immune responses. Molecular cell, 64(1), pp.105-119.
Contact
Address: Gaoke Innovation Center,Guangming District, Shenzhen
Phone: +86-755-86967710
Email: webmaster@szbl.ac.cn
Postal Code: 518132
Copyright © 2025 Shenzhen Bay Laboratory. All Rights Reserved.