
电话:+86-755-86967710
邮箱:webmaster@szbl.ac.cn

Institute of Molecular Physiology
mikexzhu(at)szbl.ac.cn
Intracellular calcium signaling, ion channels including organellar channels, membrane receptors, GPCR and G protein signal transduction, autophagy, cell death mechanisms, intracellular vesicle trafficking, lysosomes, osmosis, neurodegeneration, autism, stroke, pain, itch, inflammation, fibrosis, diabetes, fatty liver disease, skin disease, cancer
Senior Principal Investigator
Full Professor
Assistant Professor / Associate Professor
Assistant Researcher
Postdoctoral Research Associate
Postdoctoral Research Associate
Doctor of Philosophy (Ph.D.)
Master of Science ( M.S.)
Bachelor of Science ( B.S.)
Molecular and cellular mechanisms of regulation of ion homeostasis and ion channels and their dysregulation in disease. Main areas include calcium signaling, transient receptor potential (TRP) channels, acid-sensing ion channels (ASICs), and endolysosomal two-pore channels (TPCs). We study their function in neurodevelopment, neurotransmission, pain/itch, and neurodegeneration using cell and animal models. We also explore their functions in other organs such as the heart, lungs, liver, intestines, skin, and immune cells,under normal and pathological conditions.
Calcium ions are essential for cellular functions, and calcium signaling has long been a pivotal area of biomedical research. Dr. Zhu's research focuses on intracellular calcium signaling, ion channels, membrane receptors, and G protein-coupled signal transduction, with particular emphasis on the molecular mechanisms underlying cellular signaling and ion channel regulation, as well as their physiological and pathological roles. His major achievements include:
Cloning and functional characterization of mammalian canonical transient receptor potential (TRPC) channels, followed by extensive investigations into the functional regulation and physiological / pathological roles of multiple TRP channel subtypes.
Cloning of two-pore channels (TPCs) and validation of their role in mediating nicotinic acid adenine dinucleotide phosphate (NAADP)-induced calcium release from lysosomes - further demonstration that TPCs also respond to phosphatidylinositol 3,5-bisphosphate [PI(3,5)P₂] and play critical roles in diverse cellular physiological processes and diseases in subsequent studies.
Discovery that acid-sensing ion channel subtype 1a (ASIC1a) activates receptor-interacting serine/threonine-protein kinase 1 (RIPK1) via a channel-independent mechanism to mediate acid-induced cell death - elucidation of the underlying molecular pathway, with important implications in stroke and various neurodegenerative disorders.
Dr. Zhu's academic influence, expertise, and contributions have earned broad international recognition. He actively participated in the entire process of establishing the standardized nomenclature for TRP channels. He has been invited many times to deliver presentations at premier international conferences and forums such as the Gordon Research Conferences (GRC) and FASEB Science Research Conferences (SRC), and has authored over 200 publications in leading journals, including Cell, Nature, and several other Nature journals.
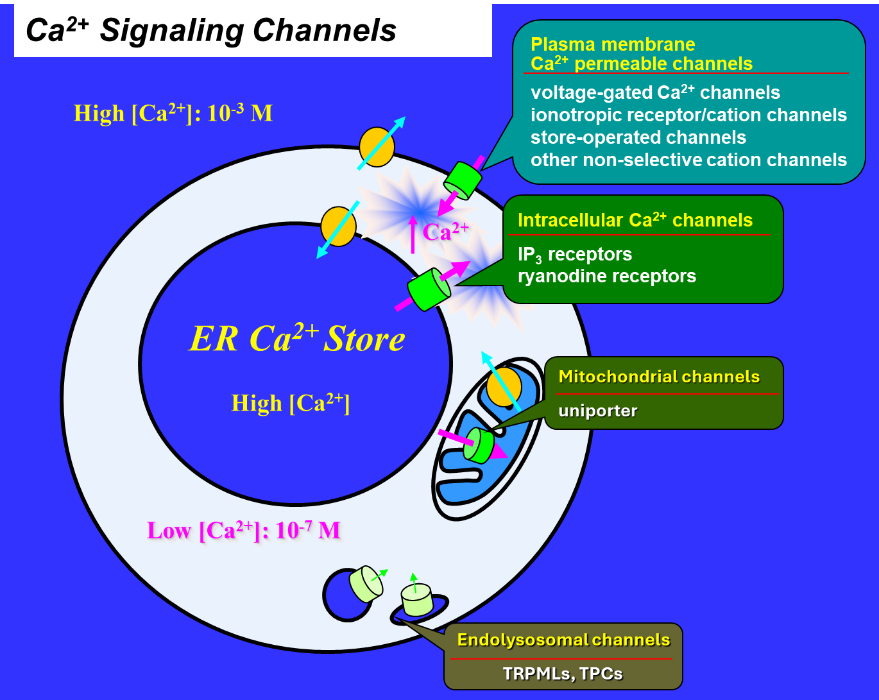
Figure 1. Principle of Ca2+ signaling. Cells maintain a very low cytosolic Ca2+ concentration ([Ca2+], ~100 nM) under resting conditions. Ca2+ concentrations in the extracellular space and in the lumens of endoplasmic reticulum (ER) and endosomes/lysosomes (collectively endolysosomes) are at millimolar levels. The opening of Ca2+-permeable channels on the plasma membrane, ER membrane, and endolysosomal membranes allows Ca2+ flow into the cytosol, causing an increase in cytosolic [Ca2+]. The Ca2+ signal plays a very important role in the life and death of the cell. Ca2+ also flows into mitochondria to regulate mitochondrial function. Major Ca2+ channel types in different cellular compartments are indicated.
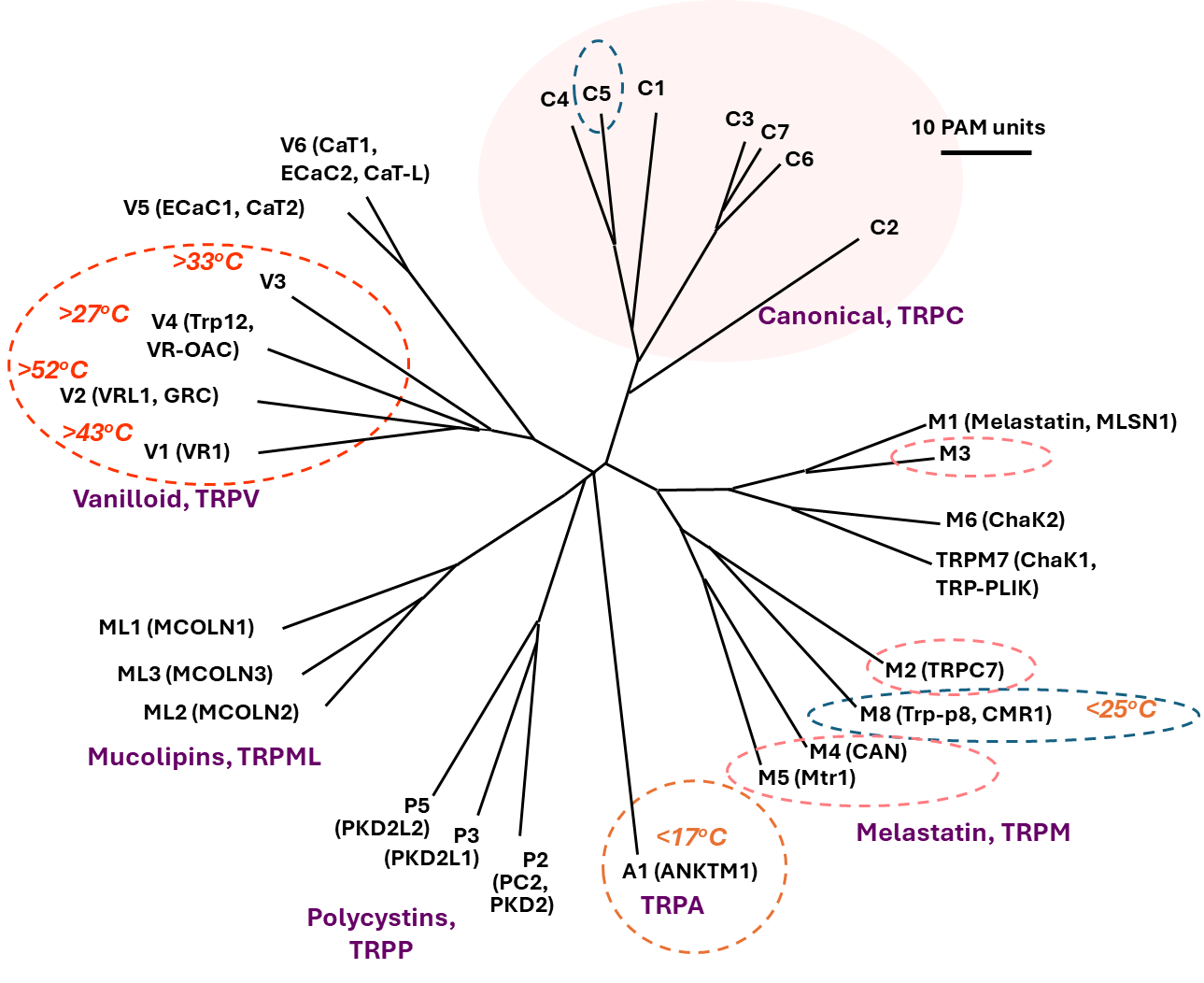
Figure 2. Transient receptor potential (TRP) ion channels. The mammalian TRP superfamily comprises 28 members, categorized into six subfamilies: TRPA, TRPC, TRPM, TRPML, TRPP, and TRPV. The canonical members, TRPCs (pink-shaded area), are closely related to the original TRP protein found in the fruit fly, which gave this superfamily its name. Dashed oval circles highlight the temperature-sensitive TRP channels. Names in parentheses are older names used before the unified nomenclature was introduced.
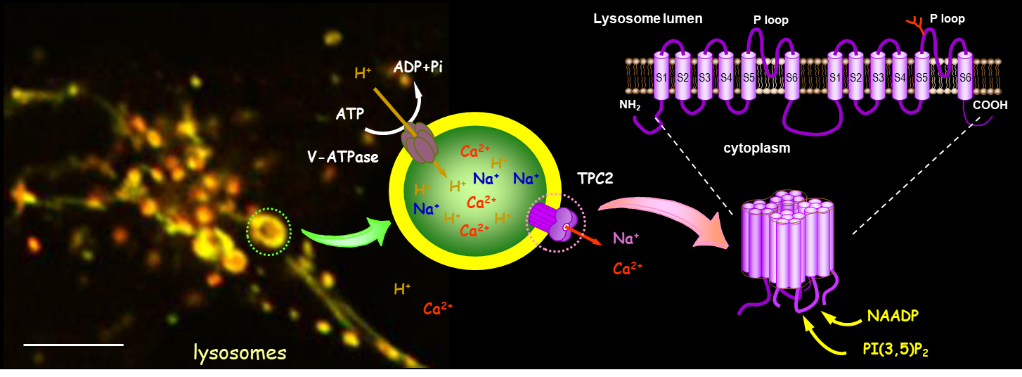
Figure 3. Two-pore channels (TPCs). TPCs (TPC1 and TPC2) are expressed on the membranes of endosomes and lysosomes. They respond to second messengers, NAADP and PI(3,5)P2, to release Ca2+ and Na+ from these acidic stores. These channels have been implicated in many important functions, such as intracellular vesicle trafficking, cholesterol transport, pigmentation, and viral infection.
1. Zhao, F., Xi, C., Li, J., Ren, K., Tang, Q., Liang, H., Yang, S., Zhu, M.X., and Cao, Z. Molecular determinant of low-voltage dependence of human Nav1.7 inactivation revealed by efficacy-based Nav1.7 selective inhibitor. Nat Commun. 2026, Article in Press. doi: 10.1038/s41467-026-69184-8.
2. Jeon, J., Moore, T.I., So, I., Freichel, M., Flockerzi, V., Birnbaumer, L., and Zhu, M.X. TRPC4 regulates limbic behavior and neuronal development by stabilizing dendrite branches through actomyosin-driven integrin activation. Proc Natl Acad Sci U S A. 122(33):e2511037122, 2025. doi: 10.1073/pnas.2511037122.
3. Guo, S.S., Gong, Y., Zhang, T.T., Su, X.Y., Wu, Y.J., Yan, Y.X., Cao, Y., Song, X.L., Xie, J.C., Wu, D., Jiang, Q., Li, Y., Zhao, X., Zhu, M.X., Xu, T.L., and Liu, M.G. A thalamic nucleus reuniens-lateral septum-lateral hypothalamus circuit for comorbid anxiety-like behaviors in chronic itch. Sci Adv. 10(33):eadn6272, 2024. doi: 10.1126/sciadv.adn6272.
4. Xiong, J., Luu, T.T.T., Venkatachalam, K., Du, G., and Zhu, M.X. Glutamine produces ammonium to tune lysosomal pH and regulate lysosomal function. Cells 12(1):80, 2022. https://doi.org/10.3390/cells12010080.
5. Gao, Y., Ma, R., Weng, W., Zhang, H., Wang, Y., Guo, R., Gu, X., Yang, Y., Yang, F., Zhou, A., Cheng, J., Chen, Z.Y., Zhu, M.X., and Li, Y. TRPV1 SUMOylation suppresses itch by inhibiting TRPV1 interaction with H1 receptors. Cell Rep. 39(11):110972, 2022. doi: 10.1016/j.celrep.2022.110972.
6. Tian, J.B., Yang, J., Joslin, W.C., Flockerzi, V., Prescott, S.A., Birnbaumer, L., and Zhu, M.X. TRPC4 and GIRK channels underlie neuronal coding of firing patterns that reflect Gq/11-Gi/o coincidence signals of variable strengths. Proc Natl Acad Sci U S A. 119(20):e2120870119, 2022. doi: 10.1073/pnas.2120870119.
7. Zhan, Q., Jeon, J., Li, Y., Huang, H., Xiong, J., Wang, Q., Xu, T.L., Li, Y., Ji, F.H., Du, G., and Zhu, M.X. CAMK2/CaMKII activates MLKL in short-term starvation to facilitate autophagic flux. Autophagy 18(4):726-744, 2022. doi: 10.1080/15548627.2021.1954348.
8. Wang, J.J., Liu, F., Yang, F., Wang, Y.Z., Qi, X., Li., Y., Hu, Q., Zhu, M.X., and Xu, T.L. Disruption of auto-inhibition underlies conformational signaling of ASIC1a to induce neuronal necroptosis. Nat Commun. 11(1):475, 2020. doi: 10.1038/s41467-019-13873-0.
9. Calcraft, P.J., Ruas, M., Pan, Z., Cheng, X., Arredouani, A., Hao, X., Tang, J., Rietdorf, K., Teboul, L., Chuang, K.T., Lin, P., Xiao, R., Wang, C., Zhu, Y., Lin, Y., Wyatt, C.N., Parrington, J., Ma, J., Evans, A.M., Galione, A., and Zhu, M.X. NAADP mobilizes calcium from acidic organelles through two-pore channels. Nature 459(7246):596-600, 2009. doi: 10.1038/nature08030.
10. Zhu, X., Jiang, M., Peyton, M., Boulay, G., Hurst, R., Stefani, E., and Birnbaumer, L. trp, a Novel mammalian gene family essential for agonist-activated capacitative Ca2+ entry. Cell 85(5):661-671, 1996. doi: 10.1016/s0092-8674(00)81233-7.
Contact
Address: Gaoke Innovation Center,Guangming District, Shenzhen
Phone: +86-755-86967710
Email: webmaster@szbl.ac.cn
Postal Code: 518132
Copyright © 2025 Shenzhen Bay Laboratory. All Rights Reserved.